Allegheny Health Network Cancer Institute invests in Elekta’s MR-linac research system
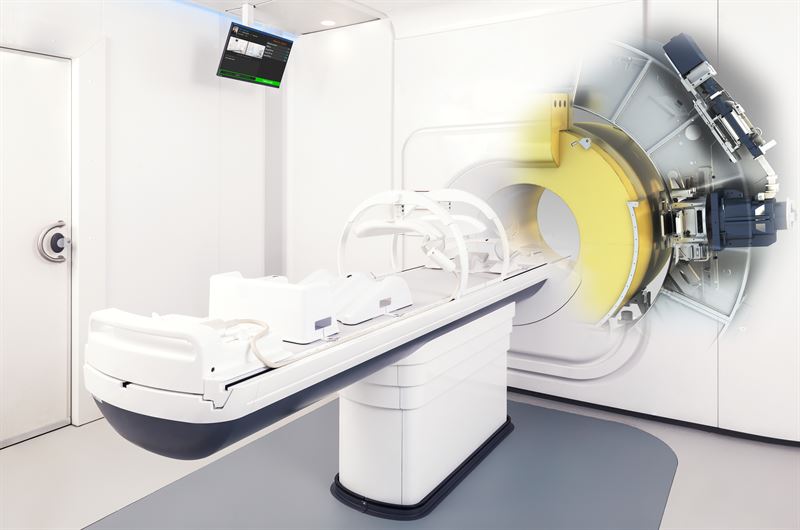
Cancer Institute to install Elekta technology, using MRI imaging to provide precise radiation delivery
PITTSBURGH, October 19, 2018 – Allegheny Health Network (AHN) Cancer Institute has entered into a research agreement with Elekta, under which AHN will utilize Elekta’s magnetic resonance radiation therapy system (MR/RT), Elekta Unity, to investigate new treatment standards, imaging protocols and treatment planning methodologies for a variety of cancers. Elekta Unity is currently pending 510(k) clearance with the U.S. Food and Drug Administration.
Six centers in the United States have ordered Elekta’s MR-linac research system and AHN will be the first center in western Pennsylvania to install Elekta Unity for clinical research. The system is currently in clinical use at a number of internationally acclaimed cancer institutes. It offers a new paradigm of precision radiotherapy, combining MRI with the radiation delivery system, supporting personalized regimens that meet the unique needs of the individual patients’ anatomy, Elekta Unity is able to visualize small soft tissue targets before and during every treatment, allowing daily adaptation of the treatment plan.
“Since 2014, Elekta has been an important factor in our ability to provide the patients we serve with cutting-edge technologies and we are pleased to expand our relationship by advancing the clinical utility of Elekta Unity,” said David Parda, MD, Chair, AHN Cancer Institute. “The AHN Cancer Institute is proud to be a leader in oncology innovation, and we look forward to leveraging our clinicians’ significant research capabilities to bring this transformative approach to cancer therapy to our patients.”
The Elekta Unity system comprises more than just cutting-edge hardware and software. It also encompasses collaborations with clinicians and researchers at leading cancer centers around the world to undertake a systematic evaluation of the system in specific cancer types and anatomies. This approach is designed to provide evidence-based protocols and workflows that optimize the use of MR/RT across defined cancer indications and within specific patient populations.
“Elekta Unity is a technological tour-de-force that was enabled through collaboration with leading researchers and clinicians who treat cancer patients every day,” said Peter Gaccione, Elekta’s Executive Vice President, Region North and Latin America. “Our innovation strategy embraces the critical insights that those on the front lines of cancer care can provide to our development efforts. Expanding this strategy to encompass the development of clinical protocols and workflows is a tangible example of how our focus on patients allows us to go beyond products and enable new treatment paradigms. We look forward to the contributions AHN will make in this endeavor.”
To learn more, visit elekta.com/MRRT.
Elekta Unity is pending 510(k) pre market clearance and not available for commercial distribution or sale in the U.S.
# # #
For further information, please contact:
Oskar Bosson, Global EVP Corporate Communications and Investor Relations
Tel: +46 70 410 7180, e-mail: Oskar.Bosson@elekta.com
Time zone: CET: Central European Time
Raven Canzeri, Global Public Relations Manager
Tel: +1 770 670 2524, e-mail: raven.canzeri@elekta.com
Time zone: EST: Eastern Standard Time
Stephanie Waite, Strategic Communications Advisor, Allegheny Health Network
Tel: +1 412 544 0848, e-mail Stephanie.Waite@highmarkhealth.org
Time zone: EST: Eastern Standard Time
About Elekta
Elekta is proud to be the leading innovator of equipment and software used to improve, prolong and save the lives of people with cancer and brain disorders. Our advanced, effective solutions are created in collaboration with customers, and more than 6,000 hospitals worldwide rely on Elekta technology. Our treatment solutions and oncology informatics portfolios are designed to enhance the delivery of radiation therapy, radiosurgery and brachytherapy, and to drive cost efficiency in clinical workflows. Elekta employs 3,700 people around the world. Headquartered in Stockholm, Sweden, Elekta is listed on NASDAQ Stockholm. www.elekta.com
About Allegheny Health Network
Allegheny Health Network, a Highmark Health Company, is a western Pennsylvania-based integrated healthcare system that serves patients from across a five state region that includes western Pennsylvania and the adjacent regions of Ohio, West Virginia, Maryland and New York. The Network’s Cancer Institute employs more than 200 physicians and 500 oncology professionals who provide a complete spectrum of oncology care at more than 20 affiliated oncology clinics, including access to state-of-the-art technologies and new therapies being explored in clinical cancer trials. The Cancer Institute has the only cancer program in the Pittsburgh region with Integrated Network Cancer Program accreditation by the American College of Surgeons Commission on Cancer, and its radiation oncology program is the largest in the country accredited by the American College of Radiology. AHN Cancer Institute is a Quality Oncology Practice Initiative certified practice, and is accredited by the Foundation for Accreditation of Cellular Therapy and National Accreditation Program for Breast Centers. AHN also has a formal affiliation with the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins, one of the nation’s 41 comprehensive cancer centers designated by the National Cancer Institute, for research, medical education and clinical services. To schedule an appointment with an AHN oncologist, please call 412.DOCTORS or visit www.ahn.org/find-a-doctor.
Tags:


