Licensing agreement in India enables advanced cancer treatment with Elekta Unity
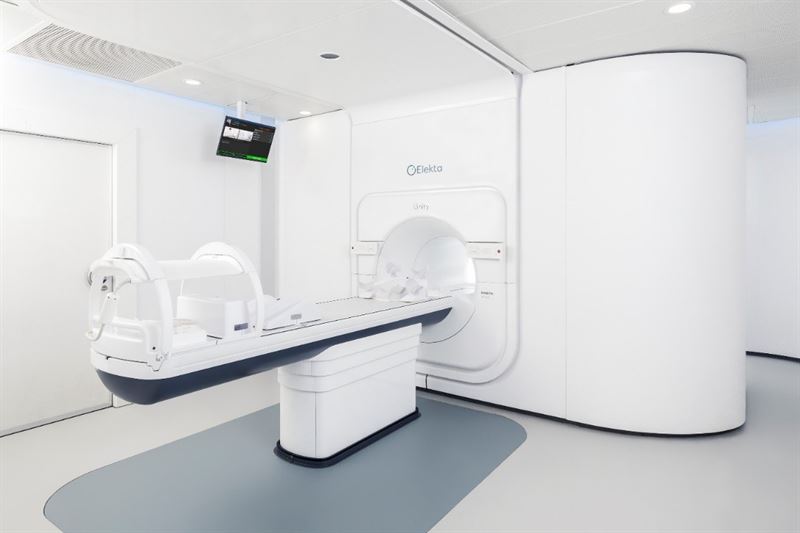
GURGAON, India, August 26, 2020 – Elekta (EKTA-B.ST) announced today that it has entered a service agreement with Healthcare Global Enterprises Ltd (HCG), India’s largest provider of cancer care. According to the agreement, Elekta will install multiple Versa HD™ and Elekta Synergy® linear accelerators (linacs), as well as an Elekta Unity MR-Linac, a radiation therapy system that allows doctors to see a tumor’s movements and its exact position during the delivery of therapeutic radiation. Also included in the agreement are Elekta’s MOSAIQ® Oncology Information System (OIS) and its Monaco® treatment planning system.
Dr. BS Ajaikumar, Chairman and CEO of HealthCare Global Enterprises, says: “We are delighted to join hands in this innovative partnership with Elekta. HCG, having a patient-centric approach and Elekta being an oncology-focused company, bringing in innovations and technology not only in linear accelerators but also in informatics and software development globally. We believe this will bring the best global standards of care to patients across India and Africa. We are also excited to be the first in India to offer radiotherapy using the MR-Linac technology from Elekta. HCG has always been a leader and a game-changer in India and emerging markets. This adds another milestone in our journey, making cancer care accessible to all.”
Gustaf Salford, Acting President and CEO at Elekta, says: “Elekta’s collaboration with HCG presents an enormous opportunity to the millions of people with cancer in India. By investing in the very best technology available, cancer patients can hope to have improved outcomes and quality of life. And as the second largest country in the world by population, we see a tremendous need for all parts of our precision radiation medicine portfolio.”
According to The Global Cancer Observatory, more than 1.15 million new cancer cases were registered in India in 2018[1]. Among cancer patients today, about 25 percent globally are treated using radiation therapy, although research suggests that the treatment would be beneficial for more than 50 percent of all cancer patients.
Manikandan Bala, Vice President and Managing Director of Elekta India, adds: “We are very pleased to develop this unique agreement together with HCG. It supports one of our business strategies of striving to increase the number of linacs to meet demand in all countries. The distribution of cutting-edge solutions to treat cancer in cities of all sizes across the country will help bridge the technology access gap. By installing our most advanced equipment, HCG will be able to offer cancer patients – both in India and the markets it serves – fewer and faster radiotherapy treatments, saving time and money for everyone. This collaboration will also include knowledge transfer across HCG’s partners in the Middle East and Africa.”
# # #
For further information, please contact:
Mattias Thorsson, Head of Corporate Communications and Public Affairs
Tel: +46 70 865 8012, e-mail: Mattias.Thorsson@elekta.com
Time zone: CET: Central European Time
Raven Canzeri, Global Public Relations Manager, Elekta
Tel: +1 770-670-2524, e-mail: raven.canzeri@elekta.com
Time zone: ET: Eastern Time
About Elekta
For almost five decades, Elekta has been a leader in precision radiation medicine. Our more than 4,000 employees worldwide are committed to ensuring everyone in the world with cancer has access to – and benefits from – more precise, personalized radiotherapy treatments. Headquartered in Stockholm, Sweden, Elekta is listed on NASDAQ Stockholm Exchange. Visit elekta.com or follow @Elekta on Twitter.
[1] Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018). Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 68(6):394-424.
https://doi.org/10.3322/caac.21492 PMID:30207593


