IRRAS strengthens position in neurocritical care by acquiring proprietary assets, including four US FDA-cleared products
- Assets complement IRRAflow® and support positioning as a leading company in the space
- Sales of the acquired products will be initiated in the US in Q3
- Acceleration of IRRAS’ product development plans and significant cost savings
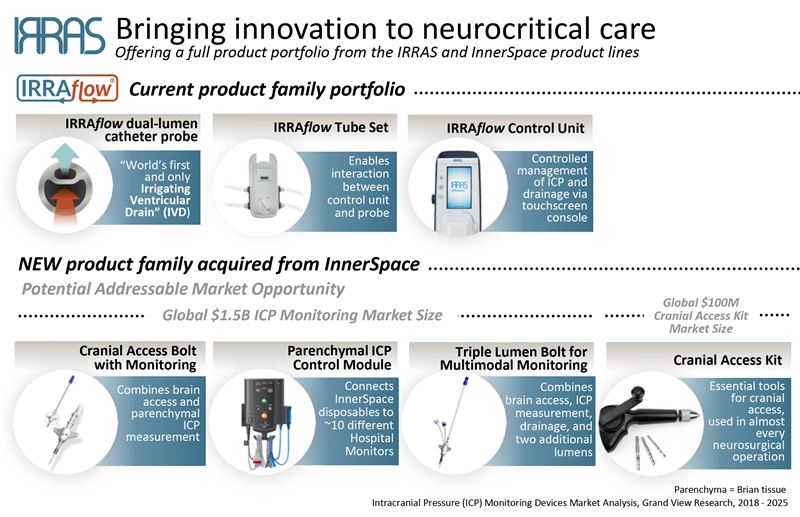
Stockholm, May 7, 2019 – IRRAS AB (Nasdaq First North Premier: IRRAS), a medical technology company focused on commercializing innovative solutions for neurocritical care, announced today it has acquired the assets of InnerSpace Neuro Solutions Inc. with immediate effect. These include proprietary single and multi-lumen cranial access bolts, parenchymal ICP (Intracranial Pressure) monitoring, and cranial access kit. The total purchase price for these assets, which are 510(K) FDA cleared in the United States but have not yet received CE Mark, was 700,000 USD.
The InnerSpace products have been used in over 2 000 patients, and their clinical utility is supported by a number of scientific publications. They complement IRRAS’ current IRRAflow product line and substantially expand the company’s available product portfolio. Sales are expected to be initiated by the IRRAS’ US commercial organization in Q3 2019.
“This is a unique opportunity for IRRAS to strengthen its position and become a leader in the neurocritical care market. These innovative and proprietary products are a perfect fit to our product portfolio. In addition, we will use established InnerSpace sales channels to introduce IRRAflow to new customers,” said Kleanthis G. Xanthopoulos, Ph.D., President & CEO.
There are clear and immediate commercial synergies that will provide IRRAS with innovative solutions across the treatment spectrum in neurocritical care. Furthermore, the acquired patents and expertise will result in accelerated timeline and significantly reduced development costs of planned IRRAflow products.
About IRRAS
IRRAS AB (Nasdaq First North Premier: IRRAS) is a publicly-traded, commercial-stage medical technology company focused on developing and commercializing innovative solutions for brain surgery.
The company’s initial product, IRRAflow, is the world’s first “irrigating ventricular drain.” Its unique mechanism of action addresses the complications associated with the current methods of managing intracranial fluid by using a dual lumen catheter that combines active irrigation with ongoing fluid drainage. Additionally, IRRAflow incorporates ICP monitoring and uses a proprietary software to regulate treatment based on desired pressure levels. IRRAflow received FDA-clearance in July 2018.
With its unique product portfolio, protected by property patents and patent applications, IRRAS is well positioned to establish a leadership position in the medical device market. IRRAS maintains its headquarters in Stockholm, Sweden, with corporate offices in Munich, Germany, and San Diego, California, USA. For more information, please visit www.irras.com.
IRRAS AB (publ) is listed on Nasdaq First North Premier. Wildeco is certified adviser of the company. Wildeco is reached at + 46 8 545 271 00 or at info@wildeco.se.
For more information, please contact:
US
Kleanthis G. Xanthopoulos, Ph.D.
President & CEO
info@irras.com
Europe
Fredrik Alpsten
CFO and Deputy CEO
+46 706 67 31 06
fredrik.alpsten@irras.com
This document is considered information that IRRAS is obliged to disclose pursuant to the EU Market Abuse Regulation. The information was released for public disclosure, through the agency of the contact person above, on May 7, 2019 at 08.00 a.m. (CET).
Tags:


