Targovax announces encouraging data in mesothelioma study combining ONCOS-102 and standard of care chemotherapy
· Preliminary data show a numerical advantage in Progression Free Survival in ONCOS-102 treated patients · Robust immune activation in the experimental group, with a positive association between immune response and clinical outcome · Combination treatment with ONCOS-102 and chemotherapy is well tolerated · Discussions ongoing with a prospective pharma collaboration partner to move forward with a checkpoint inhibitor combination study Oslo, Norway, 21 January 2020 - Targovax ASA (OSE: TRVX), a clinical stage immuno-oncology company developing oncolytic viruses to target hard-to-









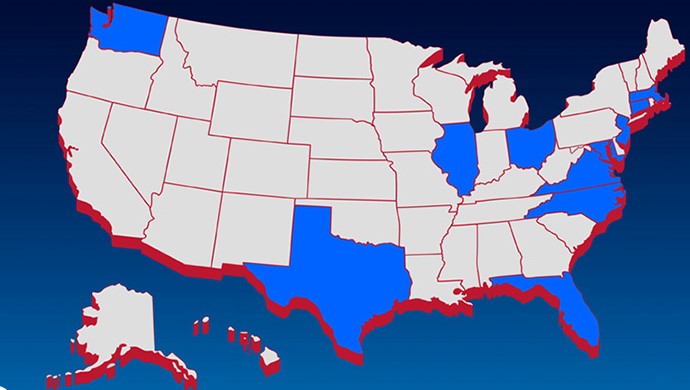 Conotoxia, Inc. continues to conquer the U.S. Its services of fast and cost-effective multi-currency money transfers are now available in further states.Conotoxia, Inc. is quickly expanding its Money Transfer service in the U.S. After the Company’s successful initial launch in Illinois last month, it is now opening to 10 more states: Ohio, Florida, New Jersey, Massachusetts, Connecticut, Virginia, Maryland, Washington, North Carolina, and Texas. The Company aims to offer its service to all 41 states in which it currently holds licenses.
Conotoxia, Inc. continues to conquer the U.S. Its services of fast and cost-effective multi-currency money transfers are now available in further states.Conotoxia, Inc. is quickly expanding its Money Transfer service in the U.S. After the Company’s successful initial launch in Illinois last month, it is now opening to 10 more states: Ohio, Florida, New Jersey, Massachusetts, Connecticut, Virginia, Maryland, Washington, North Carolina, and Texas. The Company aims to offer its service to all 41 states in which it currently holds licenses.