Update on TRAUMAKINE development
Faron Pharmaceuticals Oy (“Faron” or the “Company”) · U.S. Food and Drug Administration (FDA) approval of new HIBISCUS study protocol in patients with COVID-19 infection · Company seeking patent protection for the sequential use of IV interferon beta-1a and corticosteroids · Pipeline expansion into additional organ protection indications · CMC scale-up in progress Company announcement, 26 January 2021 at 9.00 AM (EET)Inside information TURKU – FINLAND – Faron Pharmaceuticals Oy (AIM: FARN, First North: FARON), the clinical stage biopharmaceutical company, announces


 The cooperation positions the companies to challenge how traditional business consultancies approach and deliver strategyHelsinki-based strategy advisory company Taival has today announced its partnership with strategy, design, and technology consultancy Reaktor in order to help organizations continuously drive and implement strategic and technological developments that prepare them for tomorrow’s ever-changing world.
As a part of the partnership, Taival has raised €300k in seed funding with Reaktor as the lead investor, which will
The cooperation positions the companies to challenge how traditional business consultancies approach and deliver strategyHelsinki-based strategy advisory company Taival has today announced its partnership with strategy, design, and technology consultancy Reaktor in order to help organizations continuously drive and implement strategic and technological developments that prepare them for tomorrow’s ever-changing world.
As a part of the partnership, Taival has raised €300k in seed funding with Reaktor as the lead investor, which will

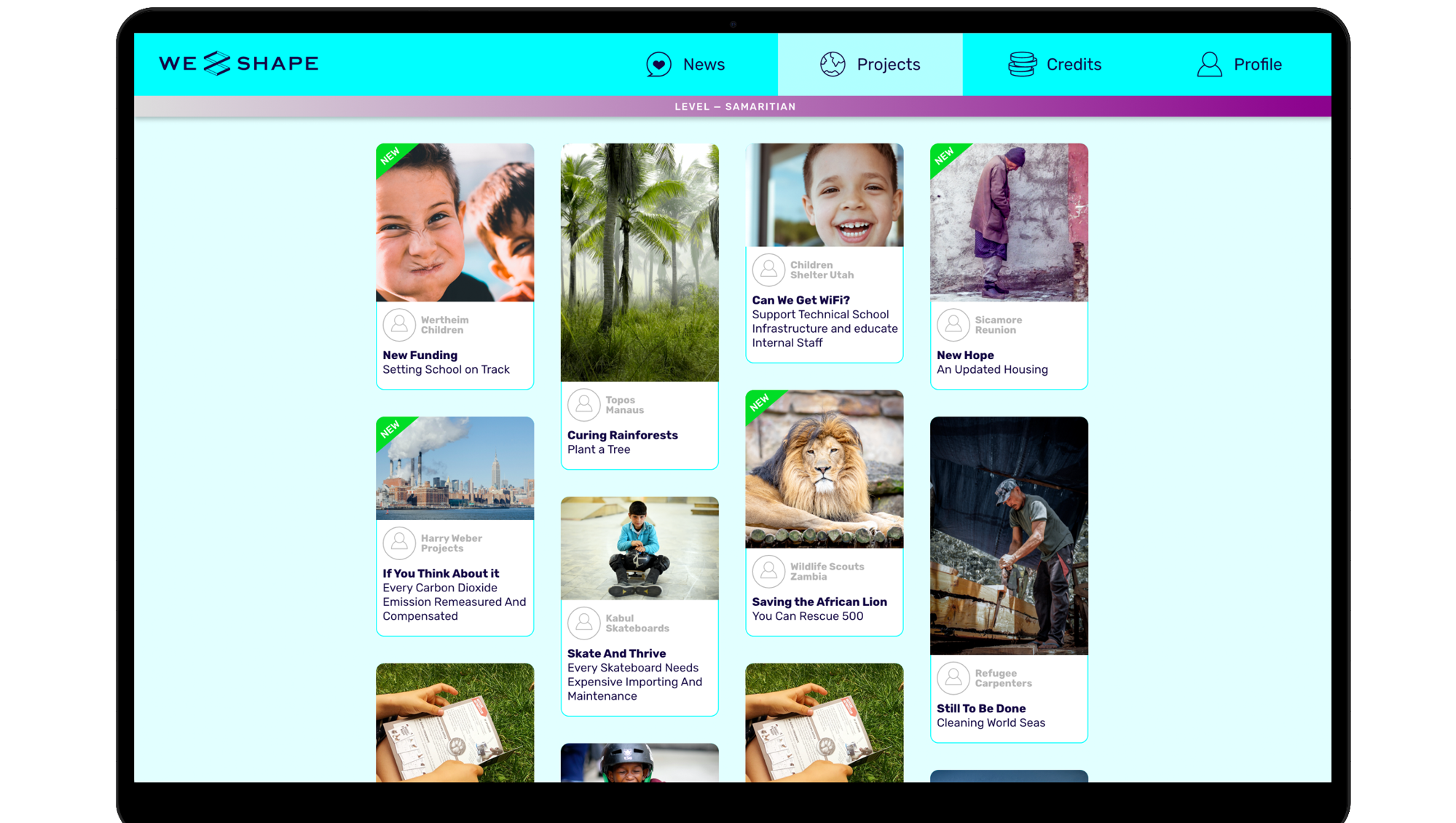 Through the CSR engagement platform WeShape.world , non-profit organizations get free access to corporate donations. Thanks to the innovative approach of involving employees in the donation process and an integrated newsfeed, nonprofits also benefit from increased visibility and direct engagement with supporters.Nonprofits can sign up for the free use of WeShape via the contact form or by emailing partnerships@weshape.world. After a simple onboarding process, organizations benefit from high reach and the ability to build
Through the CSR engagement platform WeShape.world , non-profit organizations get free access to corporate donations. Thanks to the innovative approach of involving employees in the donation process and an integrated newsfeed, nonprofits also benefit from increased visibility and direct engagement with supporters.Nonprofits can sign up for the free use of WeShape via the contact form or by emailing partnerships@weshape.world. After a simple onboarding process, organizations benefit from high reach and the ability to build





